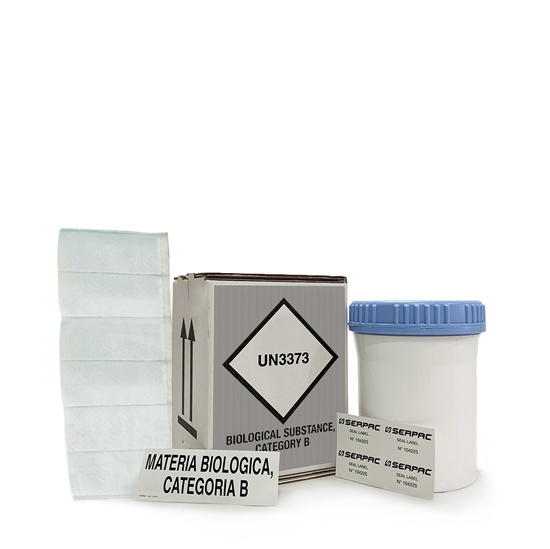

BIO-2030-SB
Complete packaging for UN3373 with 95kPa tested secondary container of 0,95 l and accessories - suitable for Covid-19
Complete packaging for UN3373 with 95kPa tested secondary container of 0,95 l and accessories - suitable for Covid-19
For the shipment of biological substances, Category B, it’s mandatory to use packagings tested and certified according to the regulations of dangerous goods transport. Italy has set some guidelines for the shipment of biological products, or better biological substances, Category B, which are mentioned in the Circolare n. 3, released on May 8th, 2003 by the Ministry of Health. However, that document is obsolete, since not aligned with some requests introduced by the new regulations.
Our BIO-2030-SB packagings, provided in kit, are tested and certified according to the packing instructions P650 ADR, P650 RID, P650 ADN, P650 IMDG, P.I. 650 IATA and therefore they are appropriate for any type of shipment. The kit includes all the necessary components to ship without any risk.
This packaging was tested and certified to contain 1 jar, certified 95 kPa (reference BIO-1000-SB).
The jar can be autoclaved at 121°C for 22 minutes (1,12 bar) and at 135°C for 11 minutes (2,1 bar) as pressure tests at 95 kPa have been successfully performed after sterilizing the jars.
With the purchase of the products, a test report is available. The report states that the packaging was tested for different primary receptacles (test tubes, bottles, vials, Eppendorf, etc.) some of them made of plastic and some made of glass.
Packaging suitable for shipment of Covid-19 (Coronavirus) swabs
| Provided | Assembled |
| In compliance with | P650 ADR, P650 RID, P650 ADN, P650 IMDG, P.I. 650 IATA |
| Capacity | 0.95 L |
| Outer dimensions | 130x130x180h mm |
| Inner dimensions | jar ⌀ 100x149h mm |
| Country of origin | Italy |
| Package | of 16 pieces |
| Content |
|---|
| Pre-printed outer cardboard with full compliance labelling for UN3373 Biological Substance, Category, rigid 95kPa from +40°C to +55°C tested secondary container of 0,95 l, certified 95kPa from +40°C to +55°C, cardboard insertion, nr. 1 absorbent pouch of 6 slots, instruction sheet, security seal. COVID-19 packaging suitable for shipping Covid-19 (Coronavirus) swabs. |
| UN3373 Biological Substance, Category B | |
| Certification | Packaging tested and certified according to packing instructions P650 and P.I. 650 |
| Compliant | with Circular n.3 of 8 May 2003 of the Ministry of Health for the transport of Covid-19 swabs. |
| Product | Material | Environmental code |
|---|---|---|
| Box | Corrugated cardboard |
PAP 20

|
| Insertion | Corrugated cardboard |
PAP 20

|
| Jar and cap | Polypropylene |
PP 5
|
| Label | Paper |
PAP 22

|
| Instruction | Paper |
PAP 22

|
| Check your district's regulations. Separate the components and confer them properly. |
Click file link to download
(Some files may be visible and downloadable only after login)
Complete packaging for UN3373 with 95kPa tested secondary container of 0,95 l and accessories - suitable for Covid-19
For the shipment of biological substances, Category B, it’s mandatory to use packagings tested and certified according to the regulations of dangerous goods transport. Italy has set some guidelines for the shipment of biological products, or better biological substances, Category B, which are mentioned in the Circolare n. 3, released on May 8th, 2003 by the Ministry of Health. However, that document is obsolete, since not aligned with some requests introduced by the new regulations.
Our BIO-2030-SB packagings, provided in kit, are tested and certified according to the packing instructions P650 ADR, P650 RID, P650 ADN, P650 IMDG, P.I. 650 IATA and therefore they are appropriate for any type of shipment. The kit includes all the necessary components to ship without any risk.
This packaging was tested and certified to contain 1 jar, certified 95 kPa (reference BIO-1000-SB).
The jar can be autoclaved at 121°C for 22 minutes (1,12 bar) and at 135°C for 11 minutes (2,1 bar) as pressure tests at 95 kPa have been successfully performed after sterilizing the jars.
With the purchase of the products, a test report is available. The report states that the packaging was tested for different primary receptacles (test tubes, bottles, vials, Eppendorf, etc.) some of them made of plastic and some made of glass.
Packaging suitable for shipment of Covid-19 (Coronavirus) swabs
| Provided | Assembled |
| In compliance with | P650 ADR, P650 RID, P650 ADN, P650 IMDG, P.I. 650 IATA |
| Capacity | 0.95 L |
| Outer dimensions | 130x130x180h mm |
| Inner dimensions | jar ⌀ 100x149h mm |
| Country of origin | Italy |
| Package | of 16 pieces |
| Content |
|---|
| Pre-printed outer cardboard with full compliance labelling for UN3373 Biological Substance, Category, rigid 95kPa from +40°C to +55°C tested secondary container of 0,95 l, certified 95kPa from +40°C to +55°C, cardboard insertion, nr. 1 absorbent pouch of 6 slots, instruction sheet, security seal. COVID-19 packaging suitable for shipping Covid-19 (Coronavirus) swabs. |
| UN3373 Biological Substance, Category B | |
| Certification | Packaging tested and certified according to packing instructions P650 and P.I. 650 |
| Compliant | with Circular n.3 of 8 May 2003 of the Ministry of Health for the transport of Covid-19 swabs. |
| Product | Material | Environmental code |
|---|---|---|
| Box | Corrugated cardboard |
PAP 20

|
| Insertion | Corrugated cardboard |
PAP 20

|
| Jar and cap | Polypropylene |
PP 5

|
| Label | Paper |
PAP 22

|
| Instruction | Paper |
PAP 22

|
| Check your district's regulations. Separate the components and confer them properly. |
Click file link to download
(Some files may be visible and downloadable only after login)